The Basics: Types of Colours
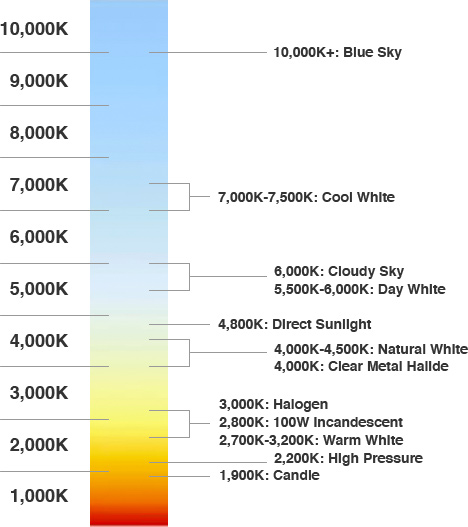
In colour we have a temperature as well. This scale is known as the kelvin colour temperature scale and detonates the colour of light based off the temperature of a black object heated to a particular temperature. As the object heats up it releases energy as electromagnetic radiation or light, but only when it gets very hot. The scale ranges from the dull orange flame of a candle at a little above 1,000K to 10,000K+ or Blue sky. Generally this type of scale is used commonly for describing the colour of a lightbulb's light.
|
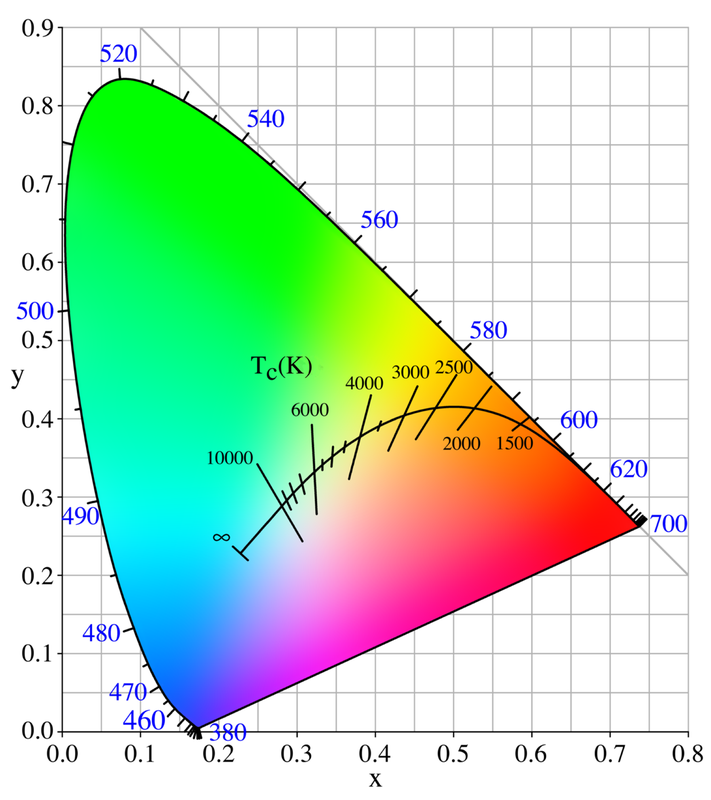
The image to the right is a colour chromaticity chart used to define a colour's chromaticity regardless of its luminosity. Chromaticity is defined by a colour's hue (literally what colour it is) and its intensity (also known as saturation). This chart and others like it generally follow the trichromacy of human vision which is assumed by most models in colour science. As the curve of the temperature scale shifts along the chart it curves towards a deep blue at infinity. In starlight we can see the blue colour in hotter faster burning stars, and yellower light from stars like our Sun.[1]
|
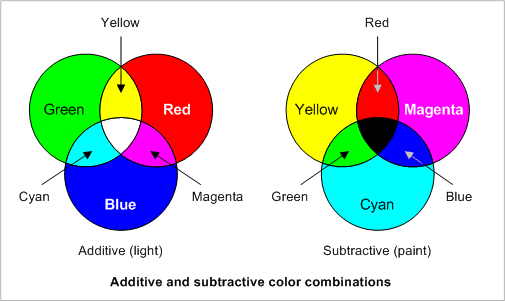
When dealing with colour there are two primary means of representation. One that we have talked about quite heavily is light. In this case the primary colours are Red, Blue, and Green. When mixed these colours add together and create white light. This type of colour mixing is known as additive. The secondary colours of the additive system are Magenta, Yellow, and Cyan. Curiously enough, these same colours are the primaries for the subtractive mixing method. The subtractive method involves pigments of colour rather than light. Pigments absorb particular wavelengths of light and reflect others. It is in this fashion that they are coloured. For example, a yellow pigment isn't actually yellow; it is in fact every colour but yellow, because it absorbs every colour and only reflects light in the yellow wavelength. This is the real reason why yellow pigments appear to be yellow to us.
Structural Colour: Blue
|
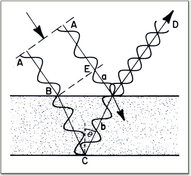
This is what the colour blue looks like. It's not a pigment that absorbs light but rather a structure that reflects light wavelengths in intervals to form blue. That reflection is the colour reflected back to us. By using an electron microscope this image of a Blue Morpho butterfly wing was captured. [2] How is this reflection of light different from pigment absorption? Well, it works by interference and reflection of the electromagnetic waveforms of light photons. Remember how when we talked about light being a wave it had a specific frequency for each colour? And each colour was defined by the distanced between each peak in the wave? |
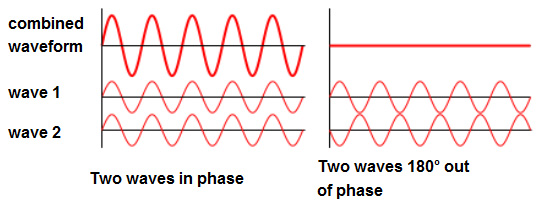
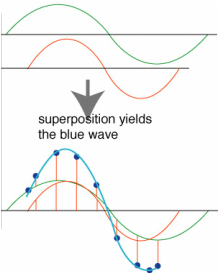
Well, this is how it works. If we want to remove a specific colour, all we have to do is reflect the same wave of light at two intervals so the peaks and troughs align in such a way that they cancel one another out.
|
This interaction is known as the superposition of a waveform. The diagram here shows two waveforms being combined to either add to one another, producing a higher energy wave or nullify one another. The higher energy wave form produced is a combination of the previous colours. To produce blue light in the case of the Blue Morpho, we need to remove the yellow light opposite it in the subtractive colour combination chart. Yellow light has a waveform of 580 nanometres (nm). So in order to nullify the waveform through superposition the spacing between the intervals of refection need to be half this length or 290 nm. The left over green and red wavelengths produce a superposition wave with natural blue in order to increase the amplitude of blue light. As you can see, the red and green waves are not perfectly aligned but still produce a single waveform in combination. In a waveform that is 100% in phase, the math is simple. Two 300 nm waves would produce a 600 nm superposition. But when the waves are out of phase to a degree (not being 180 degrees out) the wave's peaks and troughs cancel and magnify one another to a specific proportion. This proportion will determine the overall intensity of colour reflected outwards. [3]
|
The following videos are an excellent overview of structural colour and how it functions.
|
|
|
Living Colours
|
Colours are everywhere in nature and some are quite special. If you happen to have watched the video on blue hair, then you will have heard about the pigments called carotenoids. A carotenoid is an organic compound that is synthesized by both plants and a few animals. They are found in the chromoplasts and chloroplasts of these organisms and are most notably used for photosynthesis. The colour of the pigment is in the orange spectrum and through the consumption of carotenoid producing organisms the hue can be absorbed by other creatures. The flamingo is a remarkably good example of this as its feathers remain white without consuming the shrimp or blue-green algae containing the carotenoids.[4] When the flamingo eats the algae, the carotenoids travel inside the liver of the bird. Here enzymes collectively known as carotenoid oxygenases, break down the carotenoids producing the pigment that will appear in the feathers. Through this process, the absorption of colour from one animal to the next can take place. [5]
|
While carotenoids are yellow, orange, and brown; anthocyanins are the deep reds and blues found in berries and other fruits.The hue of these molecules are determined by their pH level. But what about trees? How do they work? How do they change colour in autumn?
|
In autumn there are fewer hours of sunlight and trees, at least leafy deciduous trees, have evolved to know that this means winter is coming and their leaves will die in the cold. So in preparation for the winter months, the tree begins the process of absorbing the nutrients of the leaf back into the branches of the tree. The tree does this because losing the nitrogen and phosphorus present in the green leaves every year would be far too costly for the tree's survival. So the process begins by dismantling cells and photosynthetic apparatus from the inside out. However, while the tree is moving the phosphorus and nitrogen back into the branches, any active chlorophyll molecules will begin wreaking havoc on the parts trying to transport the nutrients.
|
This occurs because the chlorophyll is still absorbing the sun's rays and producing energy. But with nowhere to send that energy, it is passed onto oxygen molecules that become highly reactive when energized. These particles will damage the leaf's nutrient return system, so something must be done. In order to counteract this, the tree will actually break down the green chlorophyll molecule into the transparent or sometime yellow chlorophyll catabolite. As a result, the orange and yellow carotenoids present in the structure of the leaf are now allowed to shine through. Some trees however, take an additional precaution against chlorophyll induced destruction. In these trees, when dismantling begins the tree produces an additional anthocyanin pigment in order to shade the chlorophyll and stop the absorption of sunlight. These pigments are red or purple in colour giving rise to the brilliant and often stunning red leaves. Unfortunately, these anthocyanins will also degrade with time as the leaf dies. With climate change effecting the length of autumn months, these pigments will degrade over time and trees will be left with more yellow leaves. [6]
Leaf colour can also be an indicator for how well the tree is doing. More information on that, and leaf colour in the video below.
References:
[1] https://en.wikipedia.org/wiki/Chromaticity
[2] http://www.scipress.org/journals/forma/pdf/1702/17020103.pdf
[3] https://en.wikipedia.org/wiki/Superposition_principle
[4] https://www.youtube.com/watch?v=5PB5l5Sk5xs
[5] https://en.wikipedia.org/wiki/Carotenoid_oxygenase
[6] https://www.youtube.com/watch?v=JWva5AaDkXw
Images:
References are beneath every image and here in order from left to right, top to bottom.
http://afremov.com/THE-PATH-TO-VICTORY-PALETTE-KNIFE-Oil-Painting-On-Canvas-By-Leonid-Afremov-Size-36-X20.html
https://store.bluerockenergyservices.com/kelvin-color-temperature-scale
https://en.wikipedia.org/wiki/Chromaticity#/media/File:PlanckianLocus.png
http://mambohead.com/wp-content/uploads/2010/11/additive-and-subtractive-color-mixing.gif
http://www.scipress.org/journals/forma/pdf/1702/17020103.pdf
https://en.wikipedia.org/wiki/Superposition_principle
http://www.webexhibits.org/causesofcolor/15.html
http://www1.kcn.ne.jp/~h-uchii/Bohr/wave.htm
http://greenlichen.com/2012/07/fact-and-fiction-real-world-cousins-of-mythical-creatures/
http://www.leamington.ca/en/index.asp
[1] https://en.wikipedia.org/wiki/Chromaticity
[2] http://www.scipress.org/journals/forma/pdf/1702/17020103.pdf
[3] https://en.wikipedia.org/wiki/Superposition_principle
[4] https://www.youtube.com/watch?v=5PB5l5Sk5xs
[5] https://en.wikipedia.org/wiki/Carotenoid_oxygenase
[6] https://www.youtube.com/watch?v=JWva5AaDkXw
Images:
References are beneath every image and here in order from left to right, top to bottom.
http://afremov.com/THE-PATH-TO-VICTORY-PALETTE-KNIFE-Oil-Painting-On-Canvas-By-Leonid-Afremov-Size-36-X20.html
https://store.bluerockenergyservices.com/kelvin-color-temperature-scale
https://en.wikipedia.org/wiki/Chromaticity#/media/File:PlanckianLocus.png
http://mambohead.com/wp-content/uploads/2010/11/additive-and-subtractive-color-mixing.gif
http://www.scipress.org/journals/forma/pdf/1702/17020103.pdf
https://en.wikipedia.org/wiki/Superposition_principle
http://www.webexhibits.org/causesofcolor/15.html
http://www1.kcn.ne.jp/~h-uchii/Bohr/wave.htm
http://greenlichen.com/2012/07/fact-and-fiction-real-world-cousins-of-mythical-creatures/
http://www.leamington.ca/en/index.asp